WATER CHEMISTRY
Assignment 1. Video Snap Shot Template - Make a copy!
Assignment 2: Colligative Properties --Link Here--
Question: Why Do You Add Salt to Boiling Water?
Why do you add salt to boiling water? There are a couple of answers to this common cooking question. Here's a look at the reason for salting water.
Answer: Usually when you're adding salt to water it's to boil the water to cook rice or pasta. Adding salt to water adds flavor to the water, which is absorbed by the food. Another reason salt is added to water is because it increases the boiling point of the water, meaning your water will have a higher temperature when you add the pasta, so it will cook better.That's how it works in theory. In reality, you would need to add 230 grams of table salt to a liter of water just to raise the boiling point by 2° C. That is 58 grams per half degree Celsius for each liter or kilogram of water. That is much more salt than anyone would care to have in their food.
Why do you add salt to boiling water? There are a couple of answers to this common cooking question. Here's a look at the reason for salting water.
Answer: Usually when you're adding salt to water it's to boil the water to cook rice or pasta. Adding salt to water adds flavor to the water, which is absorbed by the food. Another reason salt is added to water is because it increases the boiling point of the water, meaning your water will have a higher temperature when you add the pasta, so it will cook better.That's how it works in theory. In reality, you would need to add 230 grams of table salt to a liter of water just to raise the boiling point by 2° C. That is 58 grams per half degree Celsius for each liter or kilogram of water. That is much more salt than anyone would care to have in their food.
From How Water Works by Shanna Freeman on the How Stuff Works website
In its purest form, it's odorless, nearly colorless and tasteless. It's in your body, the food you eat and the beverages you drink. You use it to clean yourself, your clothes, your dishes, your car and everything else around you. You can travel on it or jump in it to cool off on hot summer days. Many of the products that you use every day contain it or were manufactured using it. All forms of life need it, and if they don't get enough of it, they die. Political disputes have centered around it. In some places, it's treasured and incredibly difficult to get. In others, it's incredibly easy to get and then squandered. What substance is more necessary to our existence than any other? Water.
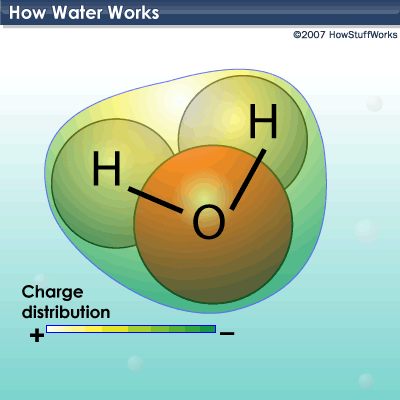
At its most basic, water is a molecule with one oxygen atom and two hydrogen atoms, bonded together by shared electrons. It is a V-shaped polar molecule, which means that it's charged positively near the hydrogen atoms and negatively near the oxygen atom. Water molecules are naturally attracted and stick to each other because of this polarity, forming a hydrogen bond. This hydrogen bond is the reason behind many of water's special properties, such as the fact that it's denser in its liquid state than in its solid state (ice floats on water). We'll look closer at these special properties later.
Water is the only substance that occurs naturally as a solid (ice), a liquid and a gas (water vapor). It covers about 70 percent of the Earth for a total of approximately 332.5 million cubic miles (1,386 million cubic kilometers) [source: U.S. Geological Survey]. If you're familiar with the lines "Water, water, everywhere, nor any drop to drink" from the poem "The Rime of the Ancient Mariner," you'll understand that most of this water -- 97 percent of it -- is undrinkable because it's saltwater (see illustration on next page). Only 3 percent of the world's water supply is freshwater, and 77 percent of that is frozen. Of the 23 percent that is not frozen, only a half a percent is available to supply every plant, animal and person on Earth with all the water they need to survive [source: National Geographic].
So water is pretty simple, right? Actually, there are a lot of things about it that scientists still don't fully understand. And the problem of making sure that enough clean, drinkable water is available to everyone and everything that needs it is anything but simple. In this article, we'll look at some of these problems. We'll also explore exactly what plants, animals and people do with water and learn more about what makes water so special.
Water Properties
The hydrogen bond between water molecules that we talked about in the first section is the reason behind two of water's unique properties: cohesion and adhesion. Cohesion refers to the fact that water sticks to itself very easily. Adhesion means that water also sticks very well to other things, which is why it spreads out in a thin film on certain surfaces, like glass. When water comes into contact with these surfaces, the adhesive forces are stronger than the cohesive forces. Instead of sticking together in a ball, it spreads out.
Water also has a high level of surface tension. This means that the molecules on the surface of the water are not surrounded by similar molecules on all sides, so they're being pulled only by cohesion from other molecules deep inside. These molecules cohere to each other strongly but adhere to the other medium weakly. One example of this is the way that water beads up on waxy surfaces, such as leaves or waxed cars. Surface tension makes these water drops round so they cover the smallest possible surface area.
Capillary action is also a result of surface tension. As we mentioned, this happens in plants when they "suck up" water. The water adheres to the inside of the plant's tubes, but the surface tension attempts to flatten it out. This makes the water rise and cohere to itself again, a process that continues until enough water builds up to make gravity begin pulling it back down.
Water's hydrogen bonds are also why its solid form, ice, can float on its liquid form. Ice is less dense than water because water molecules form crystalline structures at freezing (32 degrees Fahrenheit or 0 degrees Celsius) temperatures. The thermal properties of water are also linked to its hydrogen bonds. Water has a very high specific heat capacity, which is the amount of heat per unit mass required to raise its temperature by one degree Celsius. The energy required to raise the temperature of water by one degree Celsius is 4.2 joules per gram. Water also has a high heat of vaporization, which means that it can take a lot of heat without its temperature rising much. This plays a huge part in the climate, because it means that oceans take a long time to warm up.
Water is often known as the universal solvent, which means that many substances dissolve in it. Substances that dissolve in water are hydrophilic. This means that they are as strong or stronger than water's cohesive forces. Salt and sugar are both polar, like water, so they dissolve very well in it. Substances that do not dissolve in water are hydrophobic. This is the source of the saying "oil and water don't mix." Water's solvency is why the water that we use is rarely pure; it usually has several minerals dissolved in it.
The presence of these minerals is the difference between hard water and soft water. Hard water usually contains a lot of calcium and magnesium, but may also contain metals. Soap will not lather well in hard water, but hard water isn't usually dangerous. It can also cause lime scale deposits in pipes, water heaters and toilets.
Some of the latest controversy about water's properties lies in how ice behaves when it melts. Some scientists claim that it looks about the same as it does when it's solid, except that some of its hydrogen bonds are broken. Others claim that it forms an entirely new structure. So for all of its importance, we still don't completely understand water.
In its purest form, it's odorless, nearly colorless and tasteless. It's in your body, the food you eat and the beverages you drink. You use it to clean yourself, your clothes, your dishes, your car and everything else around you. You can travel on it or jump in it to cool off on hot summer days. Many of the products that you use every day contain it or were manufactured using it. All forms of life need it, and if they don't get enough of it, they die. Political disputes have centered around it. In some places, it's treasured and incredibly difficult to get. In others, it's incredibly easy to get and then squandered. What substance is more necessary to our existence than any other? Water.
At its most basic, water is a molecule with one oxygen atom and two hydrogen atoms, bonded together by shared electrons. It is a V-shaped polar molecule, which means that it's charged positively near the hydrogen atoms and negatively near the oxygen atom. Water molecules are naturally attracted and stick to each other because of this polarity, forming a hydrogen bond. This hydrogen bond is the reason behind many of water's special properties, such as the fact that it's denser in its liquid state than in its solid state (ice floats on water). We'll look closer at these special properties later.
Water is the only substance that occurs naturally as a solid (ice), a liquid and a gas (water vapor). It covers about 70 percent of the Earth for a total of approximately 332.5 million cubic miles (1,386 million cubic kilometers) [source: U.S. Geological Survey]. If you're familiar with the lines "Water, water, everywhere, nor any drop to drink" from the poem "The Rime of the Ancient Mariner," you'll understand that most of this water -- 97 percent of it -- is undrinkable because it's saltwater (see illustration on next page). Only 3 percent of the world's water supply is freshwater, and 77 percent of that is frozen. Of the 23 percent that is not frozen, only a half a percent is available to supply every plant, animal and person on Earth with all the water they need to survive [source: National Geographic].
So water is pretty simple, right? Actually, there are a lot of things about it that scientists still don't fully understand. And the problem of making sure that enough clean, drinkable water is available to everyone and everything that needs it is anything but simple. In this article, we'll look at some of these problems. We'll also explore exactly what plants, animals and people do with water and learn more about what makes water so special.
Water Properties
The hydrogen bond between water molecules that we talked about in the first section is the reason behind two of water's unique properties: cohesion and adhesion. Cohesion refers to the fact that water sticks to itself very easily. Adhesion means that water also sticks very well to other things, which is why it spreads out in a thin film on certain surfaces, like glass. When water comes into contact with these surfaces, the adhesive forces are stronger than the cohesive forces. Instead of sticking together in a ball, it spreads out.
Water also has a high level of surface tension. This means that the molecules on the surface of the water are not surrounded by similar molecules on all sides, so they're being pulled only by cohesion from other molecules deep inside. These molecules cohere to each other strongly but adhere to the other medium weakly. One example of this is the way that water beads up on waxy surfaces, such as leaves or waxed cars. Surface tension makes these water drops round so they cover the smallest possible surface area.
Capillary action is also a result of surface tension. As we mentioned, this happens in plants when they "suck up" water. The water adheres to the inside of the plant's tubes, but the surface tension attempts to flatten it out. This makes the water rise and cohere to itself again, a process that continues until enough water builds up to make gravity begin pulling it back down.
Water's hydrogen bonds are also why its solid form, ice, can float on its liquid form. Ice is less dense than water because water molecules form crystalline structures at freezing (32 degrees Fahrenheit or 0 degrees Celsius) temperatures. The thermal properties of water are also linked to its hydrogen bonds. Water has a very high specific heat capacity, which is the amount of heat per unit mass required to raise its temperature by one degree Celsius. The energy required to raise the temperature of water by one degree Celsius is 4.2 joules per gram. Water also has a high heat of vaporization, which means that it can take a lot of heat without its temperature rising much. This plays a huge part in the climate, because it means that oceans take a long time to warm up.
Water is often known as the universal solvent, which means that many substances dissolve in it. Substances that dissolve in water are hydrophilic. This means that they are as strong or stronger than water's cohesive forces. Salt and sugar are both polar, like water, so they dissolve very well in it. Substances that do not dissolve in water are hydrophobic. This is the source of the saying "oil and water don't mix." Water's solvency is why the water that we use is rarely pure; it usually has several minerals dissolved in it.
The presence of these minerals is the difference between hard water and soft water. Hard water usually contains a lot of calcium and magnesium, but may also contain metals. Soap will not lather well in hard water, but hard water isn't usually dangerous. It can also cause lime scale deposits in pipes, water heaters and toilets.
Some of the latest controversy about water's properties lies in how ice behaves when it melts. Some scientists claim that it looks about the same as it does when it's solid, except that some of its hydrogen bonds are broken. Others claim that it forms an entirely new structure. So for all of its importance, we still don't completely understand water.