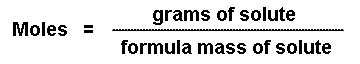Concentrations of Solutions
Theoretical yield problems - make a copy please
Assignment 21 March 3 HOMEWORK
Assignment 20 Articles Assignment
FRIDAY FEBRUARY 28:
1. Spend half of the period working on this PRACTICE QUIZ
2. TAKE AND SUBMIT THE REAL QUIZ THAT IS LINKED IN THE PRACTICE QUIZ
3. GET A MAGAZINE FROM THE BACK OF THE ROOM AND START THIS ASSIGNMENT
1. Spend half of the period working on this PRACTICE QUIZ
2. TAKE AND SUBMIT THE REAL QUIZ THAT IS LINKED IN THE PRACTICE QUIZ
3. GET A MAGAZINE FROM THE BACK OF THE ROOM AND START THIS ASSIGNMENT
Notes: Percent yield and theoretical yield
Percent Yield Practice problems --- solve then check your answers.
Percent Yield Practice problems --- solve then check your answers.
Click to set custom HTML
Download your own copy of significant figures rules
--> Assignment 17: Chapter 18 Review
Molarity (M)
Molarity is often used when more precise measurements of the concentration of solutions are needed and the solutions are being use in reactions and volumetric measurements
CLICK --> here to practice calculating molarity
Molarity is often used when more precise measurements of the concentration of solutions are needed and the solutions are being use in reactions and volumetric measurements
CLICK --> here to practice calculating molarity
-- ASSIGNMENT 13 PLEASE MAKE A COPY OF THIS PRESENTATION AND FILL IN THE BLANKS
---> Assignment 11 - Stoichiometry Stumper Presentation Project
-- > Assignment 9 Stoichiometry Review (you will complete this on hardcopy (paper) But this link includes some hints and guidelines.
-Assignment 10 Stoichiometry Stumper #1 (For Table 1 only - table 1 is closest to the orange shower)
-Assignment 10 Stoichiometry Stumper #2 (For Table 2 only - table 2 is in front of teacher desk )
-Assignment 10 Stoichiometry Stumper #3 (For Table 3 only - table 3 is lined up with part of whiteboard that projector hits)
-Assignment 10 Stoichiometry Stumper #4 (For Table 4 only - table 4 is closest to Felipe the Turtle)
--> Notes Page for How to Solve Stoichiometric Problems
-- > Presentation for Steps to Solve Stoichiometric Problems
-- Notes Page for how to set up the picket fence method
--> Presentation for Unit Conversion with Picket Fence method
--> LINK TO VIDEO EXPLAINING USING PICKET FENCE METHOD TO SOLVE STOICHIOMETRY PROBLEMS
-- Assignment 8: Paste screen shots of your completed practice problems (A,B, C, and D)
--> Assignment 7: Molar Mass Homework 2 - Due Wednesday January 22
--> Assignment 4: Molar Mass Homework -- DUE FRIDAY JANUARY 17
--> Practice problems to prepare you for quiz Friday
In chemistry, the mole is the standard measurement of amount. However, balances DO NOT give readings in moles. Balances give readings in grams.
So the problem is that, while we compare amounts of one substance to another using moles, we must also use grams, since this is the information we get from balances.
There are three steps to converting grams of a substance to moles.
So the problem is that, while we compare amounts of one substance to another using moles, we must also use grams, since this is the information we get from balances.
There are three steps to converting grams of a substance to moles.
- Determine how many grams are given in the problem.
- Calculate the molar mass of the substance.
- Divide step one by step two.
How to Calculate Molarity Using the Picket Fence Method
Stumpers Answer Key:
#1, She is lying
#2. They die shortly before reentry. However to the correct number of sigfigs the answer is 5kg so they are safe. This shows that sigfigs are a
matter of life and death!
#3. They’re in trouble (among other reasons because they don’t know how to shop). CaCl2 costs $20.00 per 50 pound bag.
#4 They’re OK to finish the race.
#1, She is lying
#2. They die shortly before reentry. However to the correct number of sigfigs the answer is 5kg so they are safe. This shows that sigfigs are a
matter of life and death!
#3. They’re in trouble (among other reasons because they don’t know how to shop). CaCl2 costs $20.00 per 50 pound bag.
#4 They’re OK to finish the race.